
Isolated tissue baths, also known as organ chambers, are tools that have been used in the discipline of pharmacology for over 150 years to evaluate tissues contractility and relaxation response upon exposure to stimuli including the addition of various agonists1. This technique was used in one of the most breakthrough scientific findings in 1980 when the endothelium pioneers, Furchgott and Zawadski, discovered the signalling molecule nitric oxide in the cardiovascular system2 for which they were awarded the 1998 Nobel Prize in physiology. The versatility of the system and the use of diverse drugs/agonists allows the investigation of different cellular pathways, to characterize of receptors and receptor signal transduction1. Understanding these aspects enabled scientists to develop numerous therapies currently used to treat millions of patients suffering from disorders such as hypertension, asthma, diabetes, heart failure, and many more.

Up to now, isolated tissue baths are still commonly used to conduct vascular reactivity studies in basic research and drug development due to their numerous benefits. The tissue organ bath system usually comes with accessories in a wide array of dimensions and shapes to handle mounting a wide variety of tissues sizes, as little as murine mesenteric and as large as porcine ileum1. Unlike other techniques which usually help to investigate each individual step such as drug-receptor affinity, signal transduction, generation of secondary messenger, tissue baths allow researchers to study the outcome from the integration of all these steps. The indispensable advantage of this technique is that living small tissues can function as a whole tissue during experimentation. The resulting physiological contraction or relaxation responses can be extrapolated to generate sound hypotheses that apply to the whole tissue which is an asset to visualize and predict how an experimental drug would work in the organism as a whole.
Isolated tissue baths maintain the integrity of the tissue for an extended amount of time by providing the desired physiological conditions in-vitro while measuring vascular reactivity response. Controlling conditions such as temperature, oxygen supply, and composition of the perfusate solution (electrolytes, nutrients, pH) allows to investigate possible changes in vascular contractility response in different environments such as hypoxia, hypercapnia, or simply normal in-vivo physiological conditions in the presence of stimuli. After the tissue of interest is delicately harvested, tissue segments of 3-4 mm wide rings are suspended between two metal stirrups placed through the lumen and connected to an isometric force transducer. The organ baths filled with the perfusate solution are pre-heated to the set temperature and the rings are washed numerous times.
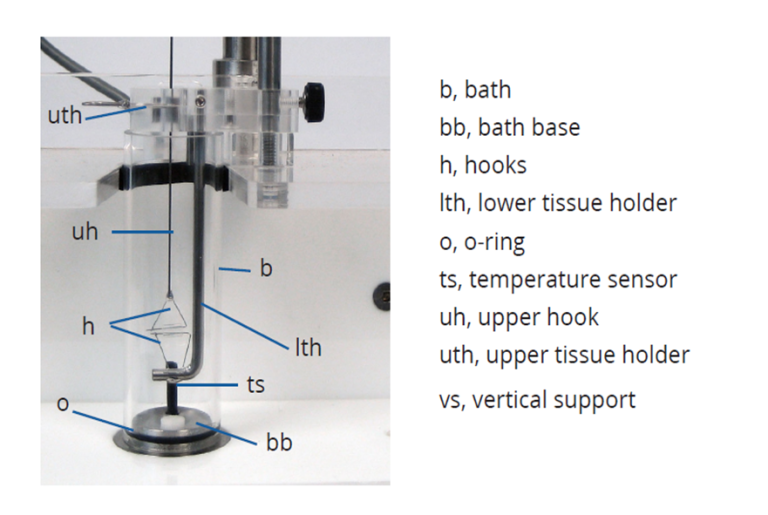
After a period of stabilization, the maximal contraction is determined with the addition of a known concentration of potassium chloride and rings are excluded if they failed to contract as it indicates damage to tissue during harvesting. After washing the potassium chloride and allowing the tissues to rest, vascular reactivity data collection is performed via force-displacement transducers interfaced with a computer using a digital data acquisition software. Commonly, vascular reactivity responses are evaluated through concentration-response curves that are performed by adding incremental concentrations of the studied drug. A myriad of additional information about pharmacological characteristics and receptor binding can be derived by using the agonist of interest in the presence or absence of an antagonist. The relative efficacy (EMAX) and potency (EC50) can be calculated and compared to responses in the same tissue by other agonists or under different environmental conditions.
Airways can be evaluated in tissue baths in order to investigate vascular reactivity of the trachea, bronchi, and bronchioles in respiratory disease. For instance, Gauthier et al in 1992 used this technique to study five generations of pre-term lambs airways to explore the effect of generation on structure-function3. Dr. Magzoub and colleagues published an interesting article in the Canadian Journal of Physiology and Pharmacology in which they evaluated the potential contracting effect of the pro-inflammatory cytokine protein leptin on isolated guinea pig tracheal smooth muscle (TSM) as well as the possible mechanism of action of this hormone through in-vitro tissue baths experiments4. Previous studies have reported that leptin plays a role in reactive lung condition such as asthma and was reported to be expressed at higher levels in the bronchi, alveoli, and proximal airways of patients with chronic obstructive pulmonary disease (COPD)5, 6. Research also uncovered the contribution of leptin to increased contraction of the coronary vascular smooth muscle7 and impairment of lung function via the leptin (Ob-R) receptor pathway8, 9. After cleaning the harvested tracheas, same length tracheal chains were assembled by suturing cartilaginous rings together. Tracheal assembled rings vascular reactivity response of leptin alone or with carbachol (CCh) was studied in organ chambers via dose-response curves. Incubation with leptin on both IgE and Ovalbumin (OVA) sensitized guinea pig TSM, which are known to mimic airway hyperresponsiveness10 in-vivo, was conducted to examine the effects of active or passive sensitization. Cotton swabs were used to strip off the epithelial later for the epithelial denudated group via scrubbing of the luminal surface to elucidate the involvement of Ob-R receptor using Ob-R antagonist.
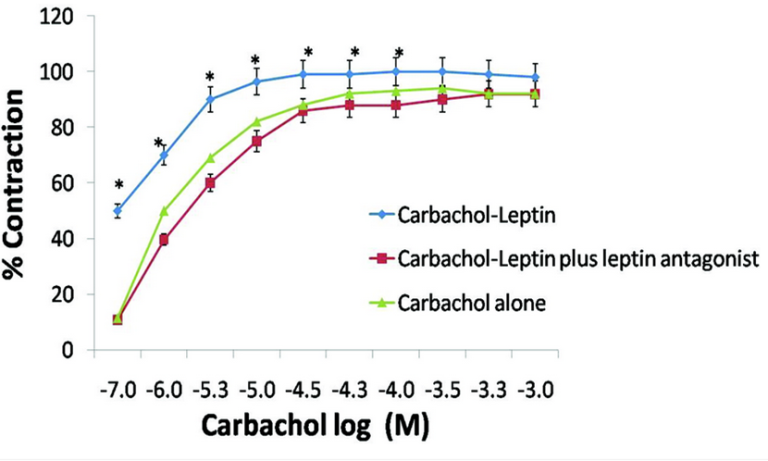
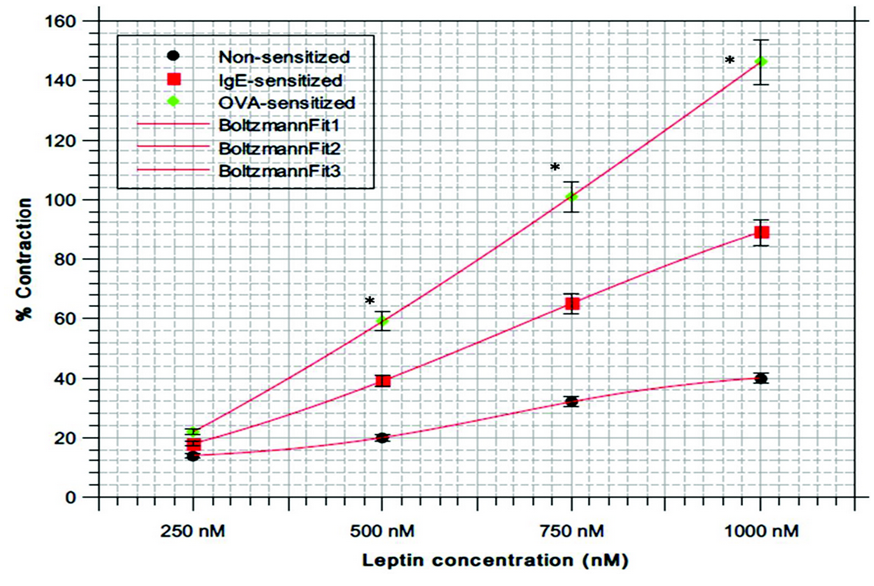
This study reported statistically significant concentration-dependent leptin contraction effects of TSM. Moreover, prior TSM incubation in leptin showed an increase of contractility upon the addition of CCh demonstrating a synergistic effect of leptin on CCh-induced contraction. In the presence of Ob-R antagonist, the mean percentage of leptin contraction decreased in both epithelium-denudated and epithelium intact trachea. This conveys the implication of the receptor Ob-R in the contracting action of leptin. Since leptin contraction did not completely vanish upon blocking Ob-R receptors, further studies are required to determine other possible complementary mechanisms involved in leptin contraction abilities. Furthermore, increased the potency of leptin-generated contraction was more evidently observed with active OVA sensitization than with IgE passive sensitization, which suggests that this phenomenon stems from the decrease of the potent vasorelaxant nitric oxide and prostaglandin E2 (PGE2). Conditions such as morbid obesity and asthma with high level of circulating leptin might display exacerbated airways reactivity due to leptin’s additive role as a modulator of TSM tone.
In conclusion, vascular reactivity studies in tissue bath are a powerful in-vitro method relevant in researching the influence of several pharmacological agents on airways smooth muscle tone in addition to understanding the pathophysiology of several respiratory disease and testing newly developed treatments in target airway tissues.
Welcome to SCIREQ’s knowledge center. You can find everything from software registration to document downloads to complete list of SCIREQ publications in this section. Everything you need to get the most out of your SCIREQ system.